Abstract
Introduction
Bone Marrow Transplant (BMT) is a potentially curative treatment for malignant and non-malignant blood disorders and has demonstrated impressive outcomes in autoimmune diseases. Prior to BMT, patients are prepared with high-dose chemotherapy alone or with total body irradiation, and both are associated with early and late morbidities, such as infertility, secondary malignancies and organ toxicity; and substantial risk of mortality. This greatly limits the use of BMT in malignant and non-malignant conditions. To address these issues, we are developing antibody drug conjugates (ADCs) targeting hematopoietic stem cells (HSCs) and immune cells to more safely condition patients for BMT.
Results
To enable simultaneous HSC and immune cell depletion for BMT we investigated targeting human CD45, a protein expressed exclusively on nearly all blood cells including HSCs. Antibody discovery campaigns identified several antibodies with sub-nanomolar affinities for human and non-human primate (NHP) CD45. We then created anti-CD45 ADCs with drug payloads including DNA-damaging, tubulin-targeting and RNA polymerase-inhibiting molecules. An ADC developed with alpha-amanitin (an RNA polymerase II inhibitor) enabled potent in vitro killing of primary human CD34+ HSCs and immune cells (40-120 picomolar IC50s).
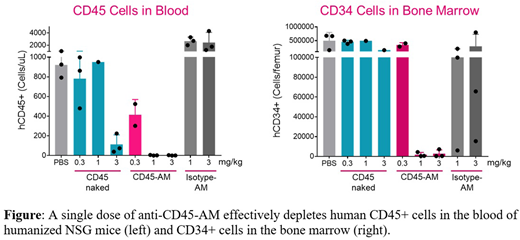
With this anti-CD45 amanitin ADC (CD45-AM), we explored depletion of HSCs and immune cells in vivo using humanized NSG mice. A single dose of 1 or 3 mg/kg CD45-AM enabled >95% depletion of human CD34+ cells in the bone marrow as assessed 7 or 14 days post-administration (Figure, n = 3/group, p values < 0.05); >95% depletion of human B-, T- and myeloid cells was observed in the periphery and bone marrow (Figure, p values < 0.05). Control non-targeting isotype matched-ADCs and anti-CD45 antibody not bearing a toxin had minimal effect on either HSC or immune cells.
In hematopoietic malignancies, an anti-CD45 ADC would ideally reduce disease burden and enable BMT. In a model of acute lymphoblastic leukemia (REH cell line, n = 10 mice/group), and 3 patient-derived models of FLT3+NPM1+ acute myeloid leukemia (n = 4-5 mice/group per model), a single dose of 1 mg/kg CD45-AM more than doubled the median survival and several mice survived disease-free (p values < 0.001).
Anti-CD45 antibodies have been investigated for BMT conditioning in patients as naked antibodies that rely on Fc-effector function to deplete lymphocytes (Biol Blood Marrow Transplant. 2003 9(4): 273-81); or as radioimmunotherapy (Blood. 2006 107(5): 2184-2191). These agents demonstrated infusion-related toxicities likely due to effector function elicited by the wild-type IgG backbone. To address this issue, we created anti-CD45 antibodies with reduced Fc-gamma receptor binding that prevented cytokine release in vitro and in humanized mice. As BMT will likely require fast clearing ADCs to avoid depleting the incoming graft, we also created fast-half-life CD45-AM variants with a t½ of 8-15 hours in mice. To determine the safety and pharmacokinetic properties of regular and fast half-life Fc-silent variants in an immune-competent large animal we tested these in cynomolgus monkeys. Single doses (3 mg/kg, iv, n = 3/group) of fast and regular half-life Fc-silent unconjugated anti-CD45 antibodies were both well tolerated in cynomolgus monkeys and displayed pharmacokinetic properties suitable for BMT.
Conclusion
These results demonstrate that targeting CD45 with an amanitin ADC results in potent in vitro and in vivo human HSC and immune cell depletion. This new CD45-AM ADC also significantly reduced disease burden in multiple leukemia models. Our results indicate Fc-silencing may avoid infusion-related toxicities observed with previous CD45 mAbs. An alpha-amanitin ADC targeted to CD45 may be appropriate for preparing patients for BMT since we hypothesize it may i) be non-genotoxic; ii) effectively deplete both HSC and immune cells; iii) avoid bystander toxicity, due to amanitin's poor cell permeability as a free toxin; and iv) kill cycling and non-cycling cells, the latter being necessary for effective HSC depletion. As our anti-CD45 ADCs are cross-reactive, we are currently investigating their HSC and immune cell depletion activity in vivo in NHPs to enable further preclinical development of these transplant conditioning agents.
Palchaudhuri:Magenta Therapeutics: Employment, Equity Ownership, Patents & Royalties; Harvard University: Patents & Royalties. Pearse:Magenta Therapeutics: Employment, Equity Ownership, Patents & Royalties. Proctor:Magenta Therapeutics: Employment, Equity Ownership. Hyzy:Magenta Therapeutics: Employment, Equity Ownership. Aslanian:Magenta Therapeutics: Employment, Equity Ownership. McDonough:Magenta Therapeutics: Employment, Equity Ownership. Sarma:Magenta Therapeutics: Employment, Equity Ownership. Brooks:Magenta Therapeutics: Employment, Equity Ownership. Bhat:Magenta Therapeutics: Employment. Ladwig:Magenta Therapeutics: Employment, Equity Ownership. McShea:Magenta Therapeutics: Employment, Equity Ownership. Kallen:Magenta Therapeutics: Employment, Equity Ownership. Li:Magenta Therapeutics: Employment, Equity Ownership. Panwar:Magenta Therapeutics: Employment, Equity Ownership, Patents & Royalties. Dushime:Magenta Therapeutics: Employment, Equity Ownership. Sawant:Magenta Therapeutics: Employment, Equity Ownership. Adams:Magenta Therapeutics: Employment, Equity Ownership. Falahee:Magenta Therapeutics: Employment, Equity Ownership, Patents & Royalties. Lamothe:Magenta Therapeutics: Employment, Equity Ownership. Gabros:Magenta Therapeutics: Employment, Equity Ownership. Kien:Magenta Therapeutics: Employment, Equity Ownership. Gillard:Magenta Therapeutics: Employment, Equity Ownership. McDonagh:Magenta Therapeutics: Employment, Equity Ownership, Patents & Royalties. Boitano:Magenta Therapeutics: Employment, Equity Ownership, Patents & Royalties. Cooke:Magenta Therapeutics: Employment, Equity Ownership, Patents & Royalties.
Author notes
Asterisk with author names denotes non-ASH members.